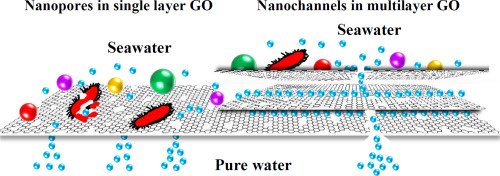
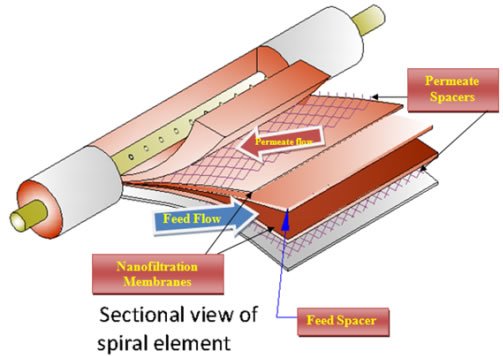
Some researchers have investigated the softening of groundwater using NF systems. All of the results gave good data. Retentions higher than 90% were found for multivalent ions, whereas monovalent ions were about 60–70%. NF membranes with pellet softening and granular activated carbon for softening water were compared. Although all methods had good results, NF membranes had several advantages from the point of health and lower investment costs. The advantage of NF softening is that a smaller stream of water can be softened because NF membranes remove essentially all hardness cations. In municipal water treatment works, it is not necessary to reduce hardness to very low values, and therefore the use of NF membranes allows for side-stream treatment. This involves sending only a portion of the flow to be softened to the membranes, and permeate is then blended with the bulk flow stream to obtain a target blended value. In contrast, precipitated lime softening cannot reduce hardness to below approximately 50 mg/L CaCO3, and therefore side-stream treatment is generally not practical.